Terumo Aortic Receives Prestigious King’s Award for Innovation

Terumo Aortic today announced that it has received the prestigious King’s Award for Enterprise in the Innovation category for its innovative bespoke solutions for the treatment of complex aortic disease using a combination of surgical and endovascular technologies. The award particularly recognises the innovation behind the Thoracoflo™ and Custom Thoraflex™ Hybrid devices, which exemplifies Terumo […]
Terumo Aortic announces US FDA Breakthrough Device Designation for Fenestrated TREO®

Terumo Aortic today announced that the US Food and Drug Administration (FDA) has granted Breakthrough Device Designation for the Fenestrated TREO® Abdominal Stent- Graft system. The FDA’s Breakthrough Device Designation program is intended to fast-track the regulatory review process for certain medical technologies and device-led combination products that satisfy certain criteria; specifically, these include providing […]
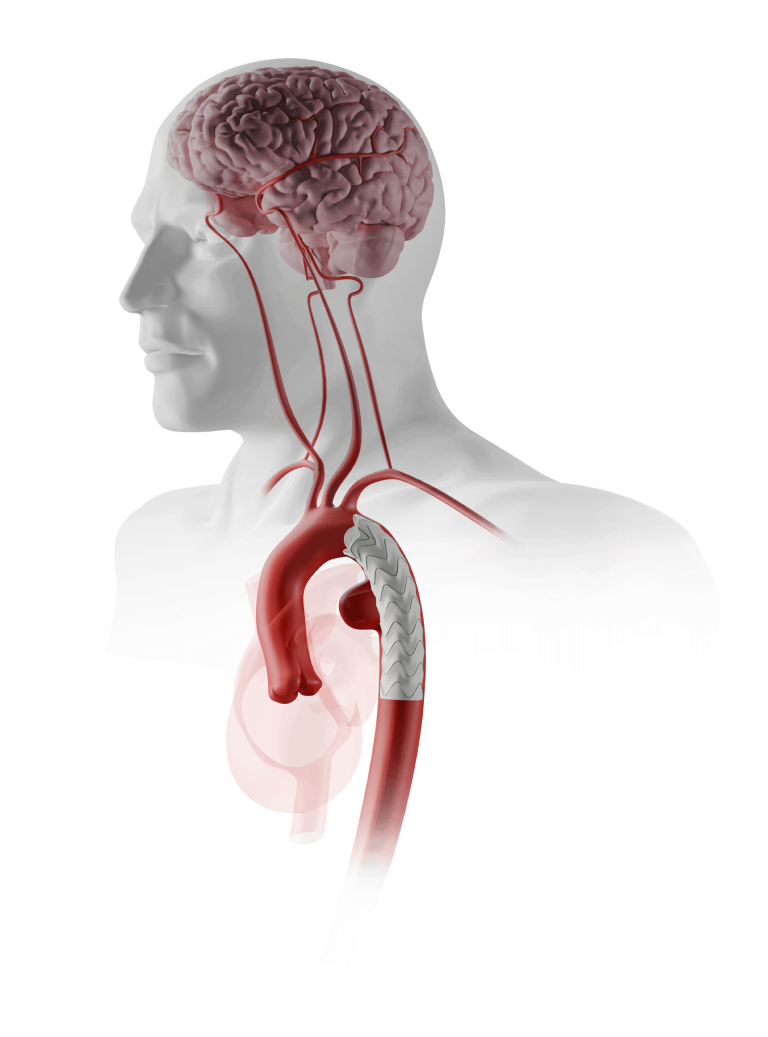
Terumo Aortic announces launch of the Rapidlink™ pivotal IDE study in the United States and Europe

Terumo Aortic today announced enrolment of the first patient in the RapidLink™ pivotal Investigational Device Exemption (IDE) study in the US and Europe*. The study is designed to evaluate the safety and effectiveness of the RapidLink™ device for the repair or replacement of supra-aortic vessels during open surgical repair of aortic disease involving the thoracic […]
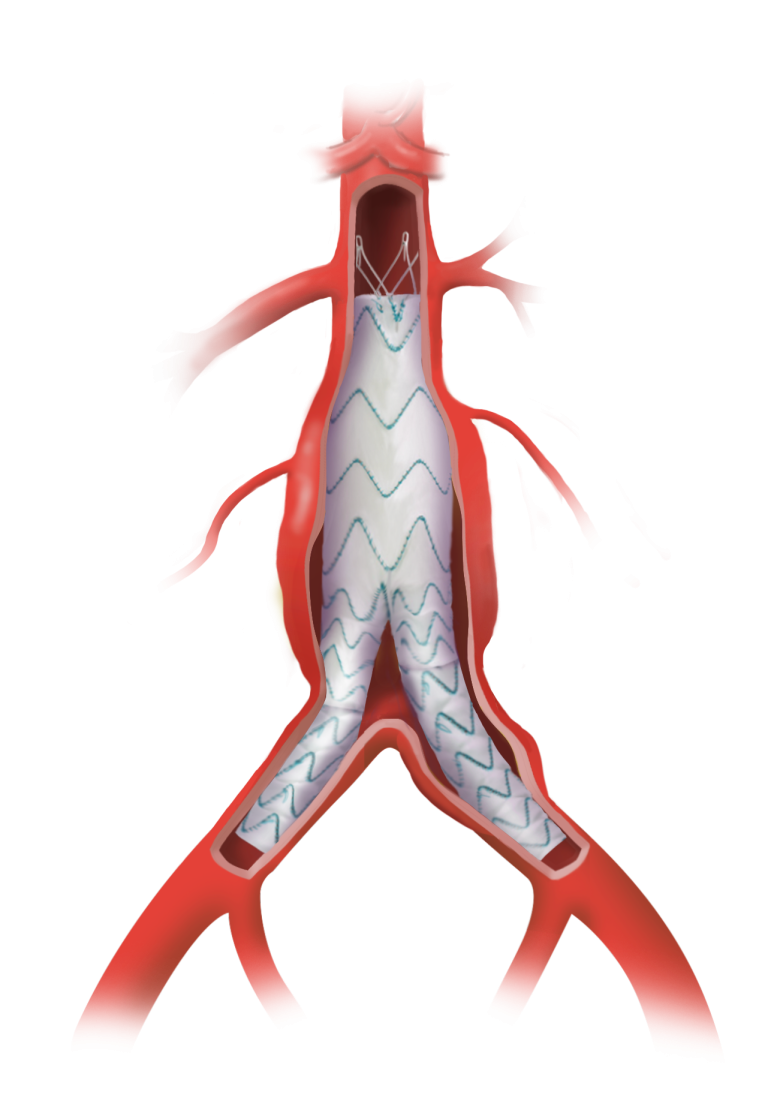
Terumo Aortic announces launch of the Fenestrated TREO® pivotal IDE study in the United States

The study is designed to evaluate the endovascular repair of juxtarenal and suprarenal aortic aneurysms using the Fenestrated TREO® Abdominal Stent-Graft System. Getinge’s iCast® covered stent system will be used as a bridging stent in this clinical study*. Achieving the first enrolment in this FDA approved IDE is a major milestone that brings us one […]
Terumo Aortic and Bentley Announce Collaboration on Groundbreaking Clinical Study

Left to right: Sebastian Büchert (CEO Bentley) and Erik Pomp (CEO Terumo Aortic) Today, Terumo Aortic, a global medical device company dedicated to developing solutions for aortic disease, and Bentley, a leading global manufacturer of balloon-expandable covered stents, announced their partnership in a clinical study in the United States. The objective is to obtain US […]
Major milestone for hospitals using Terumo Aortic’s Thoraflex™ Hybrid device as CMS establishes new DRG 209 for complex aortic arch procedures

This change will take effect on October 1, 2025, and is a significant advancement for hospitals treating patients with Thoraflex™ Hybrid in frozen elephant trunk (FET) procedures. For the last three years, hospitals using the Thoraflex™ Hybrid device have benefited from a New Technology Add-On Payment (NTAP), first granted by CMS in 2021, to help […]
Terumo Aortic Awarded Aortic Medical Devices Company of the Year

This incredible recognition reflects the trust we have built with our customers and peers across the industry – and the many nominations submitted by dedicated subscribers of MedTech Outlook Europe. This achievement marks another significant milestone for Terumo Aortic and stands as testament to our ongoing commitment to innovation, quality, and excellence in aortic care, […]
Materialise Medical – 3D Players Podcast

In this episode of the 3D Medical Players podcast, your hosts Pieter Slagmolen and Sebastian De Boodt are joined by special guest Jamie McCarte, Head of Global Custom Devices at Terumo Aortic, to discuss exactly that. Tune in now to hear his insights on: How personalized solutions are transforming the treatment of aortic diseases The […]
Terumo Aortic Receives Prestigious King’s Award for Innovation

Terumo Aortic is honoured to receive the highly prestigious King’s Award for Enterprise in the Innovation category. This very special award is in recognition of the company’s custom solutions programme for treating complex aortic disease (subject to local regulatory approval). The King’s Awards for Enterprise, previously known as The Queen’s Awards for Enterprise, represent the […]
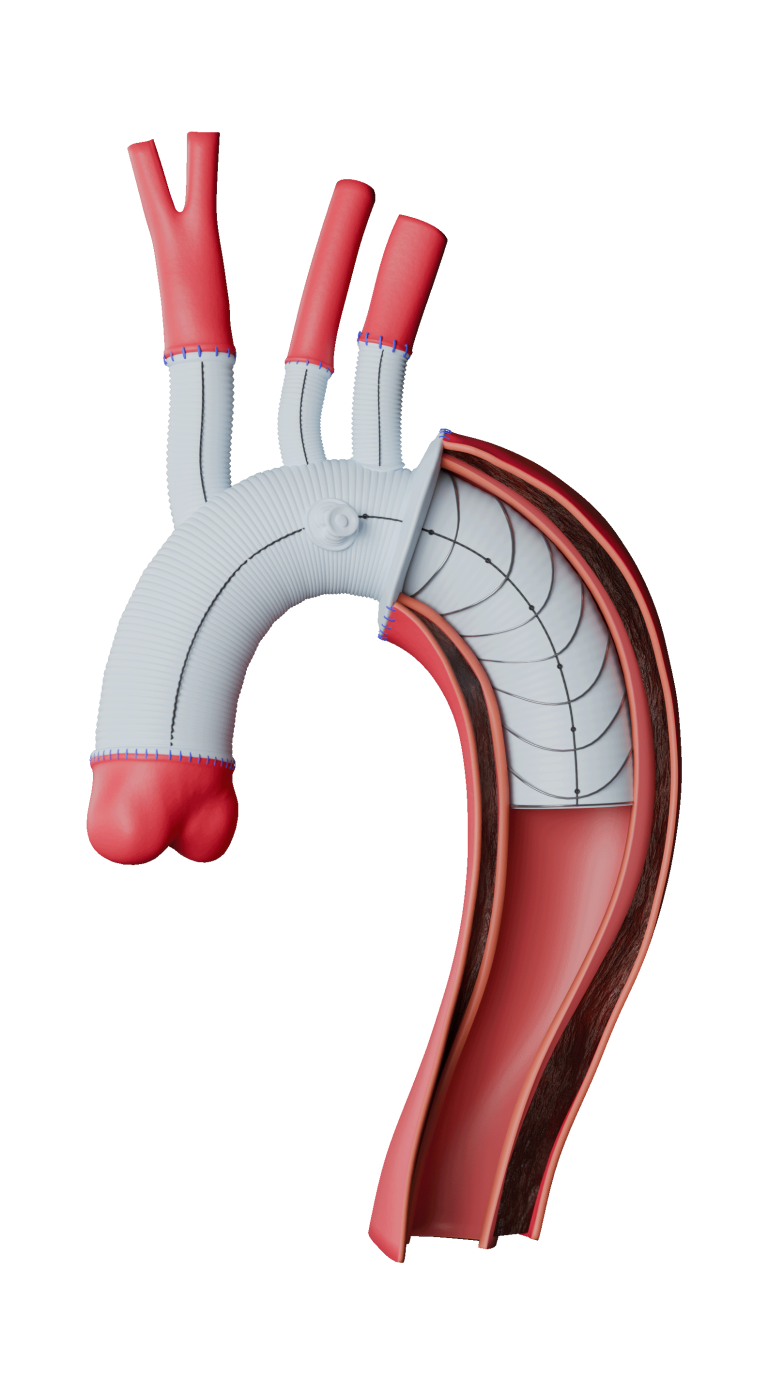
Terumo Aortic announces PMDA approval for Thoraflex Hybrid in Japan

Thoraflex Hybrid is the global market leader in Frozen Elephant Trunk (FET) repair and has well established clinical outcomes. Thoraflex Hybrid is a single use medical device combining a Gelweave polyester graft with a Nitinol self-expanding stent graft and is indicated for the open surgical repair or replacement of damaged or diseased vessels of the […]