Thoraflex Hybrid is the global market leader in Frozen Elephant Trunk (FET) repair and has well established clinical outcomes.
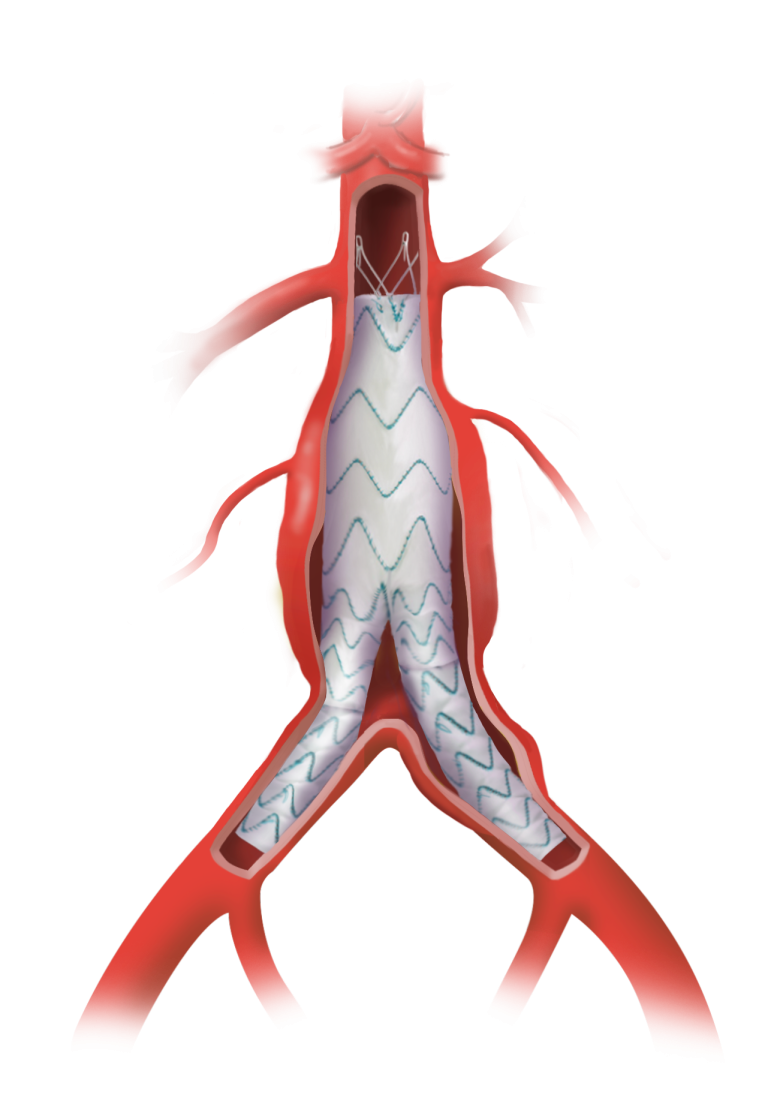
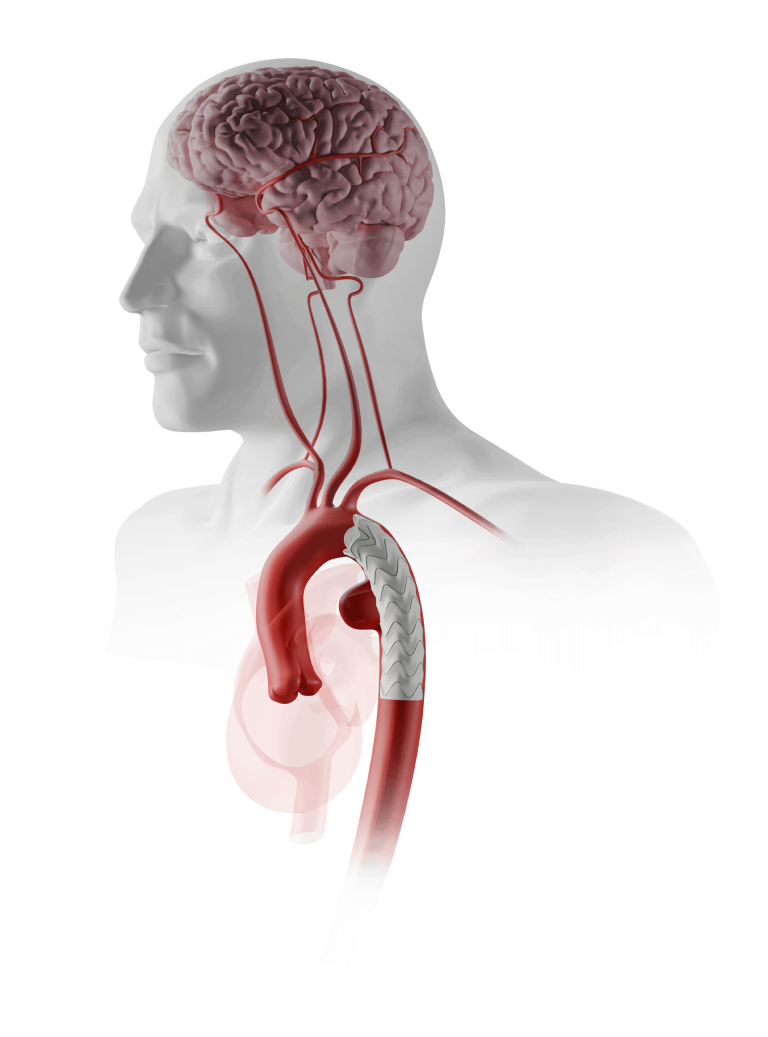
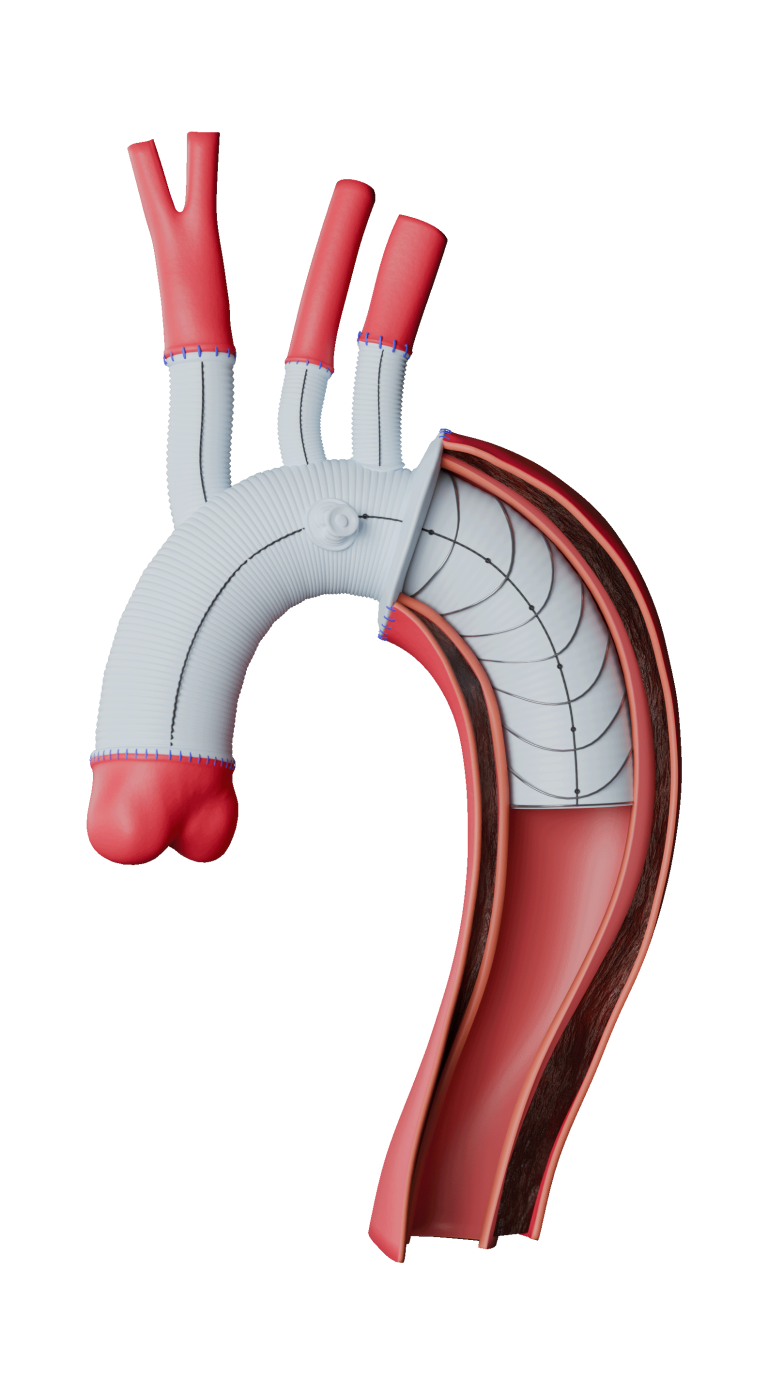
Thoraflex Hybrid is a single use medical device combining a Gelweave polyester graft with a Nitinol self-expanding stent graft and is indicated for the open surgical repair or replacement of damaged or diseased vessels of the aortic arch and descending aorta with or without involvement of the ascending aorta in cases of aneurysm and/or dissection.
Kotaro Yoshida, Senior Vice President of Asia Pacific added: “The PMDA approval of Thoraflex Hybrid is a fantastic opportunity for Terumo Aortic in Japan. This innovative hybrid device complements our open surgical graft and endovascular portfolio currently available in Japan; the approval of this device will contribute significantly in the delivery of our commitment to providing solutions for every aorta.”
Thoraflex Hybrid received CE Mark approval in 2012 and FDA approval in 2022 with more than 13,000 devices sold around the world. This device is integral to Terumo Aortic’s market-leading portfolio of surgical, endovascular and hybrid devices to treat every segment of the aorta.