Terumo Aortic today announced that the US Food and Drug Administration (FDA) has granted Breakthrough Device Designation for the Fenestrated TREO® Abdominal Stent- Graft system.
The FDA’s Breakthrough Device Designation program is intended to fast-track the regulatory review process for certain medical technologies and device-led combination products that satisfy certain criteria; specifically, these include providing a more effective treatment or diagnosis of life-threatening or irreversibly debilitating diseases or conditions. The program aims to provide patients and healthcare professionals with timely access to important breakthrough medical devices by accelerating their development, assessment and review.
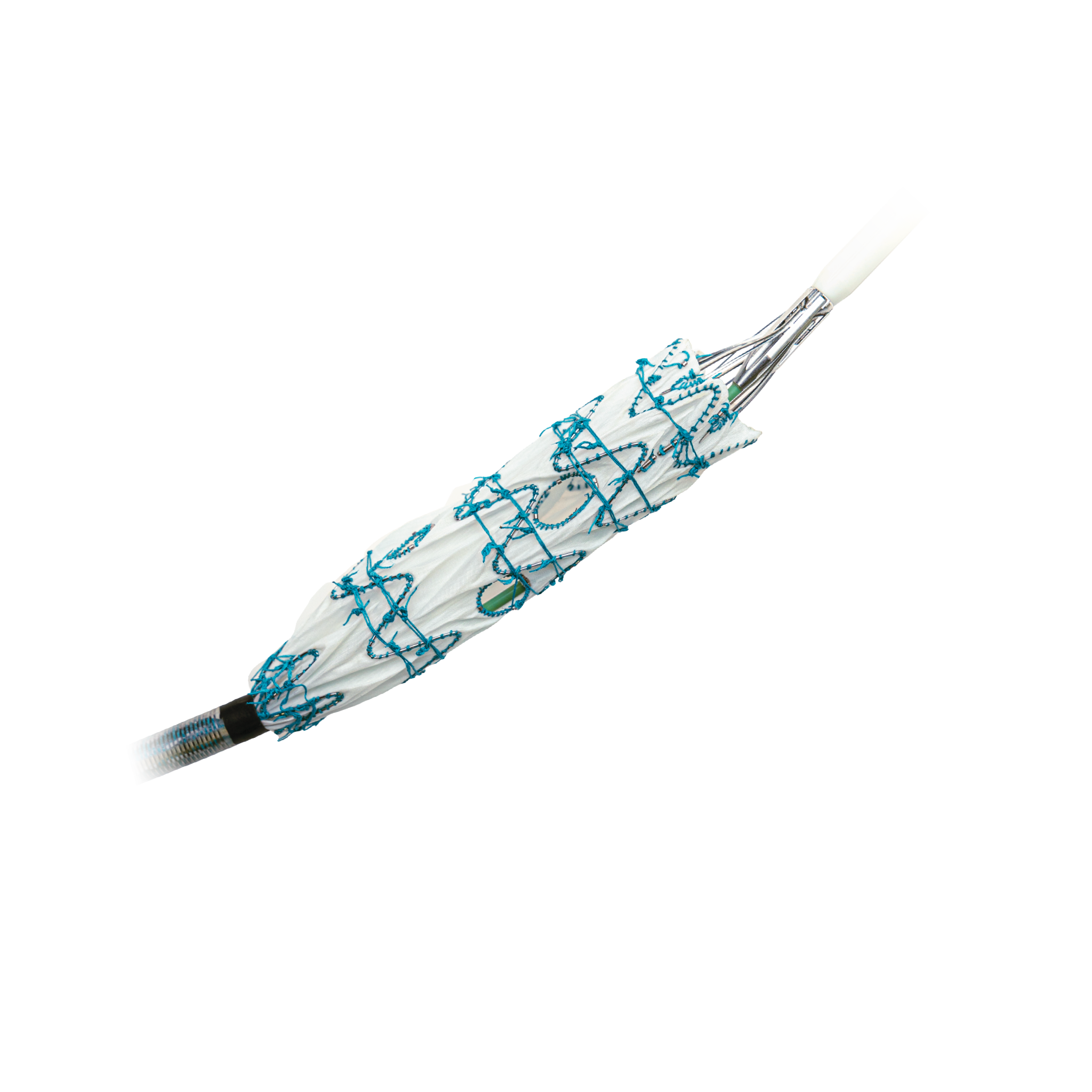
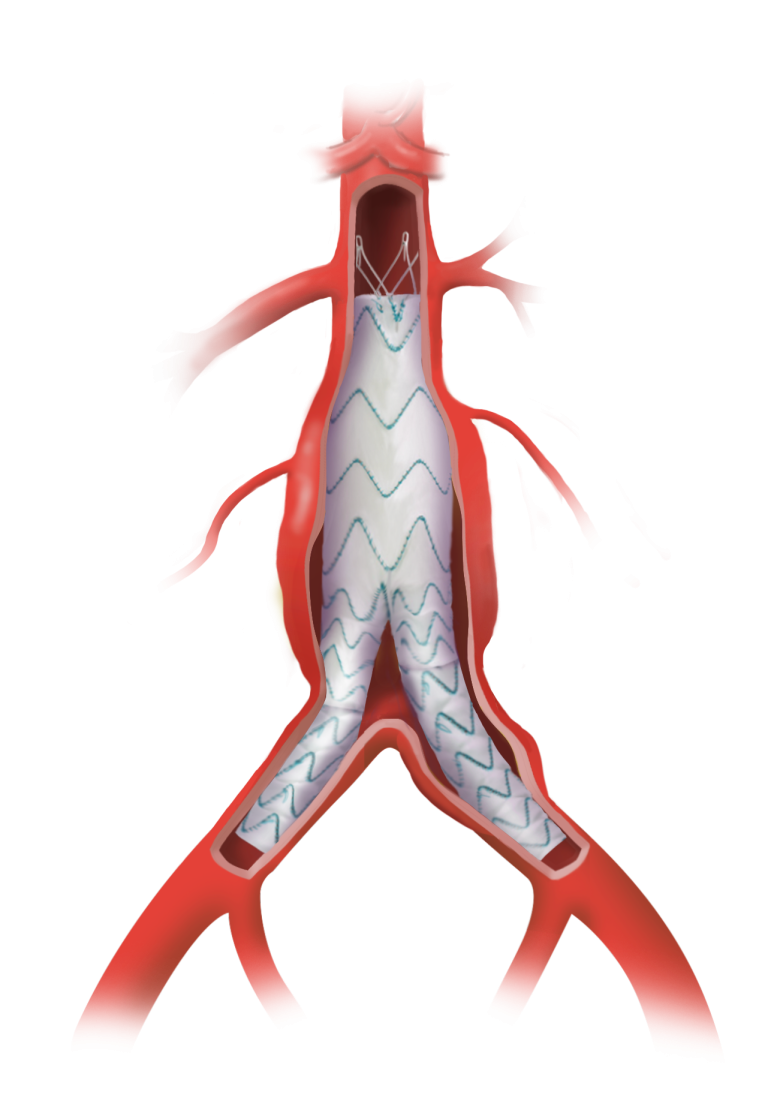
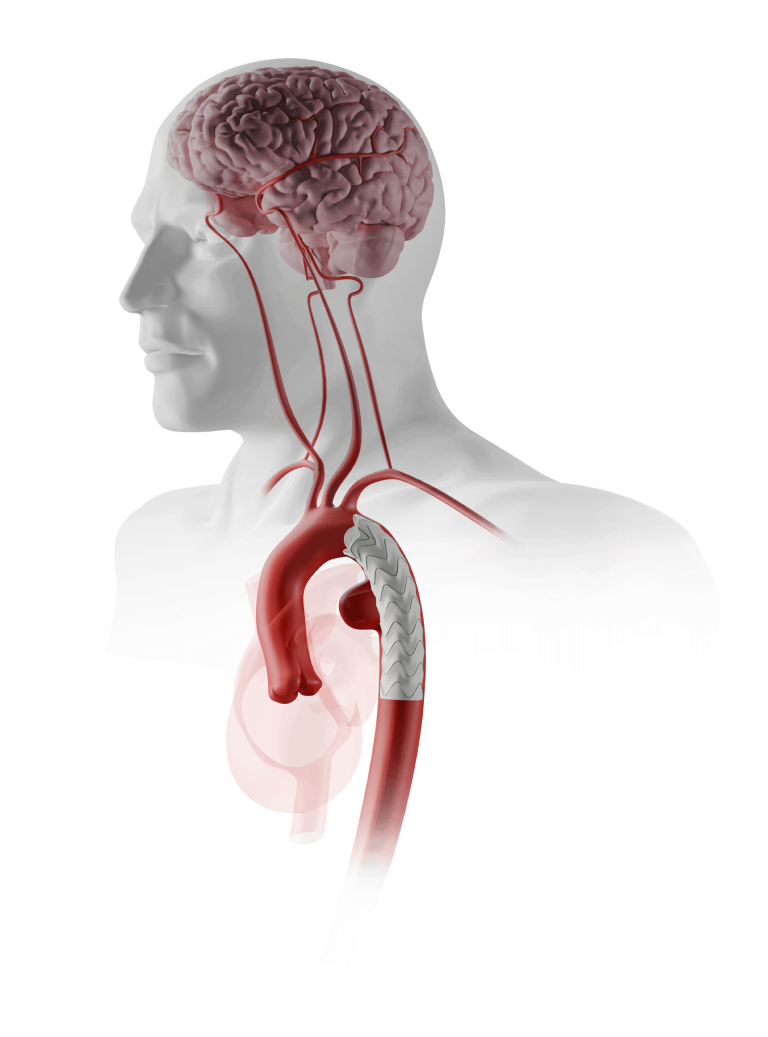
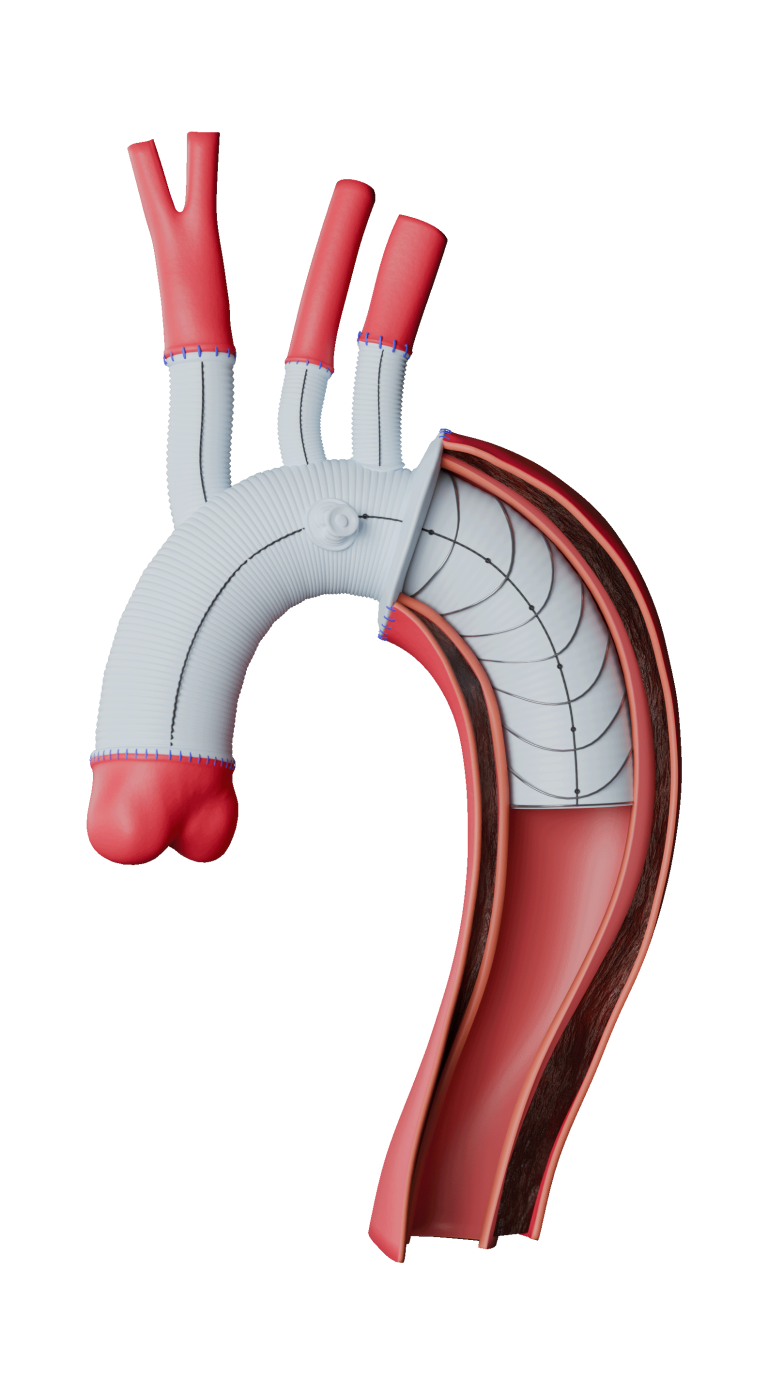
Fenestrated TREO® is a custom-made endovascular stent-graft system developed to treat complex abdominal aortic aneurysms (AAA). The device is intended for the endovascular repair of juxtarenal and suprarenal (pararenal/paravisceral) abdominal aortic and aorto-iliac aneurysms, supporting treatment in cases where standard off- the-shelf devices may not provide an adequate anatomical fit.
Jim Rushworth, President of Terumo Aortic commented: “The FDA’s Breakthrough Device Designation for Fenestrated TREO® is very encouraging, and we look forward to close collaboration with the FDA as we work to advance treatment options for patients with complex abdominal aortic aneurysms.”
Jeffrey Mifek, Global Vice President of Clinical & Medical Affairs added: “This designation supports the potential for US physicians to treat patients whose complex anatomy or clinical conditions makes traditional endovascular devices or open surgery unsuitable – addressing a significant unmet need. Fenestrated TREO® offers a tailored, minimally invasive solution designed for challenging anatomies, including the ability to incorporate multiple fenestrations precisely aligned to a patient’s unique arterial branching pattern. This level of customization helps address the needs of patients with complex vessel configurations that off-the shelf devices can’t accommodate.”