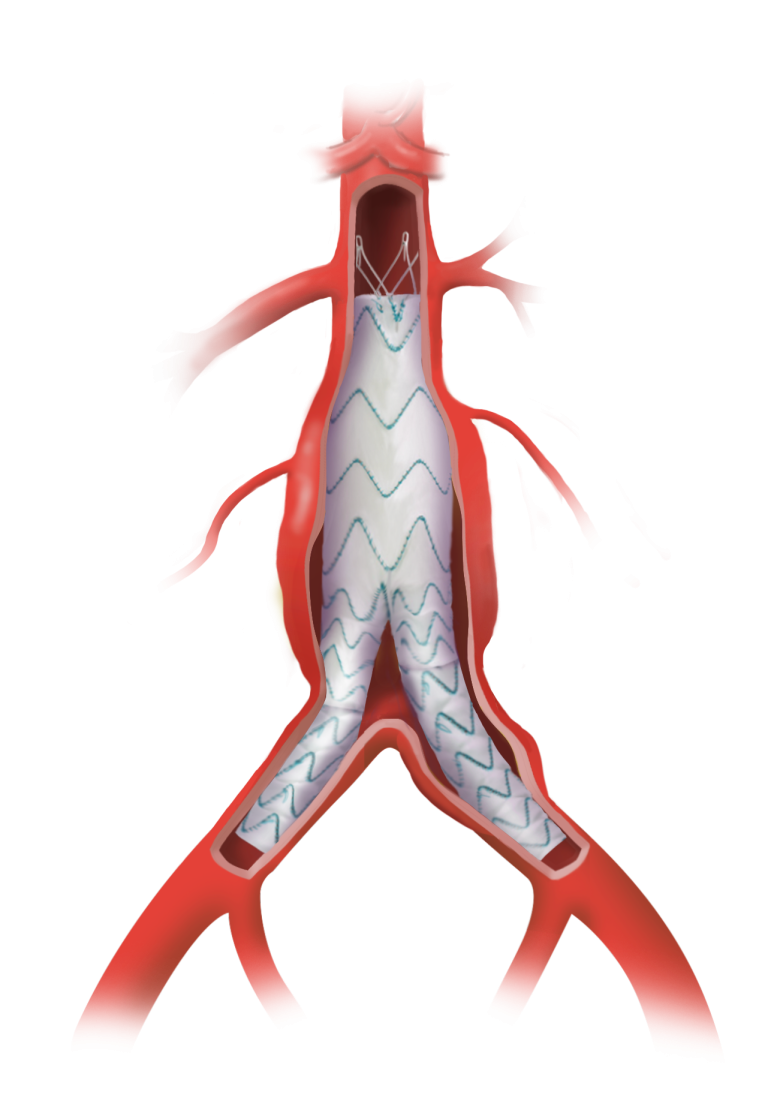
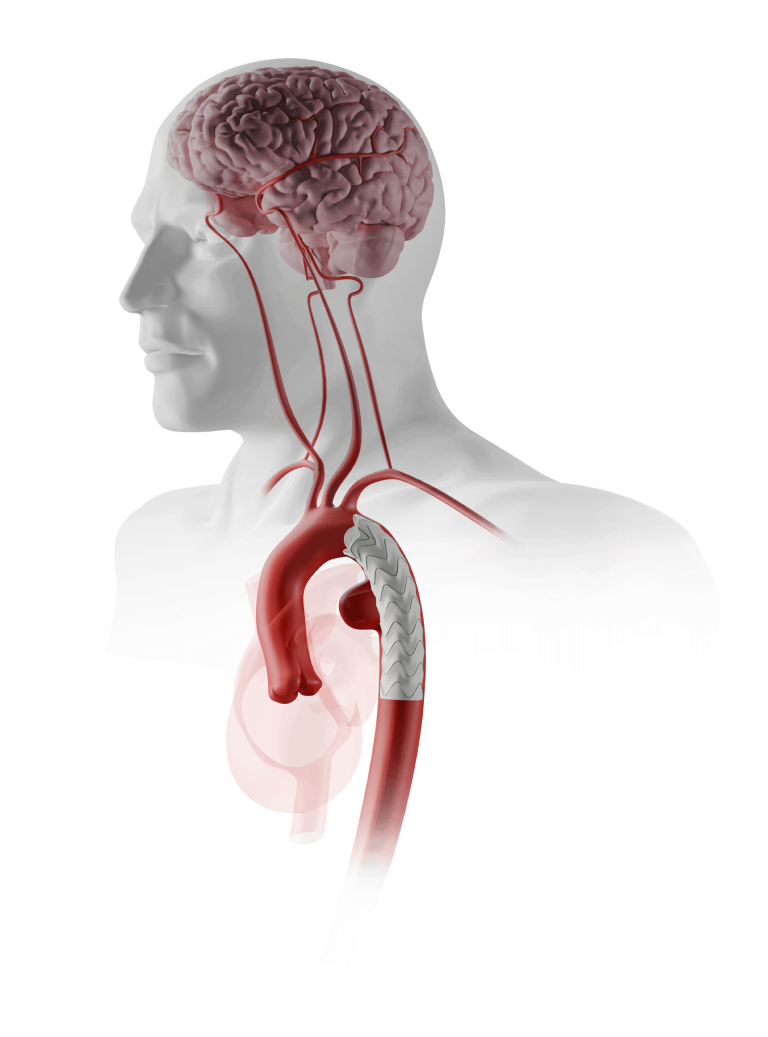
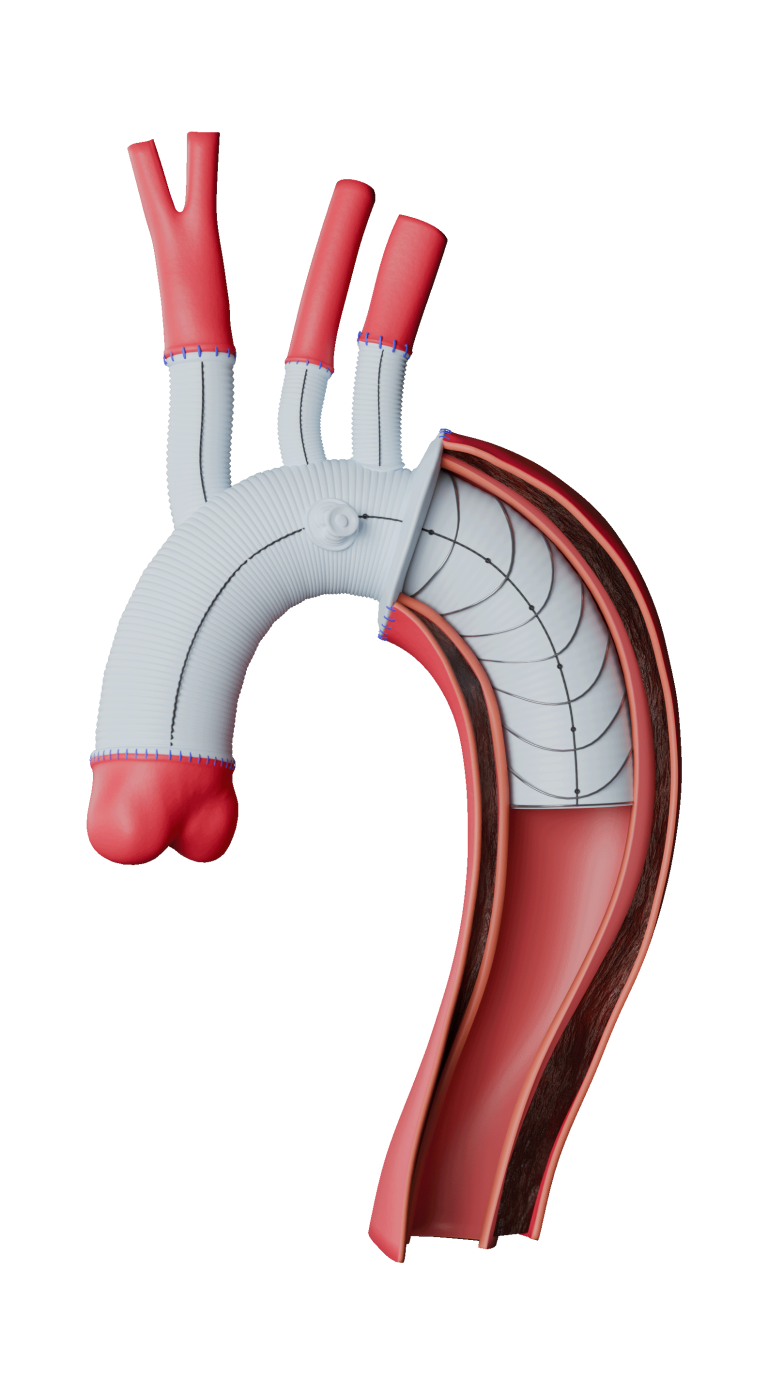
Terumo Aortic announces US FDA Breakthrough Device Designation for Fenestrated TREO®

Terumo Aortic today announced that the US Food and Drug Administration (FDA) has granted Breakthrough Device Designation for the Fenestrated TREO® Abdominal Stent- Graft system. The FDA’s Breakthrough Device Designation program is intended to fast-track the regulatory review process for certain medical technologies and device-led combination products that satisfy certain criteria; specifically, these include providing […]